Malliavin Thérèse
Biophysique et BiochmieInformations
Education
1988 Ecole Polytechnique, Palaiseau, France
1992 PhD Thesis, Université d’Orsay, France
2000 Habilitation à Diriger les Recherches, Université d’Orsay, France
Career
since 2022: researcher at Laboratoire de Physique et Chimie Théorique, CNRS and Université de Lorraine
since October 2015: Directeur de Recherches at CNRS
2006-2022: Chargé de recherches then Directeur de recherches CNRS in the Structural Bioinformatics Unit, Institut Pasteur
1998-2005: Chargé de recherches CNRS in Laboratoire de Biochimie Théorique (IBPC, Paris)
1993-1997: Chargé de recherches CNRS at Centre de Biochimie Structurale (Montpellier)
1992: Postdoc, Institut of Bioorganic Chemistry Shemyakin-Ovchinnikov (Moscow)
Computational geometry for the determination of biomolecular structures
During the last decades, structural biology allowed to incredibly improve the information on the biological objects, from single biomolecules to molecular assemblies, and to get a better description of the physics underlying the interactions between molecules. Somehow, structural biology can be considered as the description of the geometric properties of biomolecules, as biomolecular structures are determined from the measurements of angles and distances between molecule atoms. These parameter values can be determined by: Nuclear Magnetic Resonance (NMR), fluorescence/FRET, mass spectrometry coupled to cross-linking or analysis of protein sequence alignment. Up to now, one of the most used method to optimize the protein conformation given these geometric restraints, was based on the repeated use of simulated annealing (SA) algorithm, followed by extensive statistics. As the SA algorithm does not provide a guarantee of global minimality of the function, and is quite sensitive to the sparsity and to the lack of redundancy in the distance restraints, we explore the efficiency of another class of methods, derived from operations research and computational geometry, where a branch-and-pruning algorithm permits in principle the full exploration of the whole solution space. This algorithm already gave promising results on small examples of biomolecules, and the purpose of the project would be to extend them to real-life systems including: protein structures determination from NMR distance restraints, homology models or positioning of protein domains within molecular assemblies using cross-linking restraints.
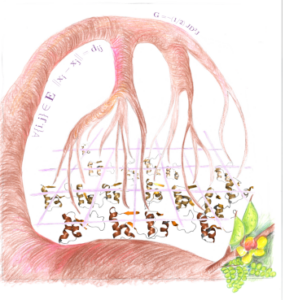
Simple description of the distance geometry approach
Molecular modeling of botulinium toxins
Although the botulinum neurotoxins (BoNTs) are among the most toxic compounds found in nature, their molecular mechanism of action is far from being elucidated. A key event is the conformational transition due to the acidification of the interior of synaptic vesicles, and leading to the translocation of the BoNT catalytic domain into the neuronal cytosol. No much is known on the structural aspects of this translocation. I am studying it using molecular dynamics simulations starting from X-ray crystallographic structures.
Last update: September 2022
Biophysique et Biochmie
Plugin is not configured correctly or shortcode parameters are not valid, please check the documentation


